Source: The Conversation – USA – By Matthew Cordes, Associate Professor of Chemistry and Biochemistry, University of Arizona

Spiders are among Earth’s most resourceful predators, nabbing prey by any means necessary. Orb weavers spin webs for capture. Wolf spiders ambush on the ground at night. Almost all spiders use venom when they hunt.
But each spider’s venom is a cocktail of ingredients as varied as their hunting behavior. Some venom ingredients can harm people, while others do not. As a result, only a few spiders threaten human health, while most are harmless and even beneficial for pest control.
In the United States, spiders that are dangerous to people include the brown recluse, which carries a necrotic toxin that destroys tissue, and the black widow, which has a special neurotoxin that damages nerve cells.
But how do these toxins work? My laboratory, in collaboration with my colleague Greta Binford, has been studying venom toxins for over two decades. In newly published research led by my former student Alexandra Sundman, we captured the structure of the recluse toxin made by the six-eyed sand spider, a relative of the brown recluse that is found in Chile. Our findings provide new clues for developing new treatments for spider bites.

Ansie Dippenaar-Schoeman/crabspider via iNaturalist, CC BY-SA
Mowing down the cell’s surface
The toxin in recluse venom is an enzyme, which is a protein that makes certain chemical reactions go faster.
The recluse toxin binds to the surface of cells and scoots along it like a lawn mower, clipping the heads off molecules on this surface. While working in my lab, my former student Dan Lajoie discovered that the toxin transforms these surface molecules into unusual ring structures. When the immune system attacks these damaged and fragile cells, it can lead to widespread tissue death called necrosis.
For reasons researchers still don’t understand, these toxins cause necrosis in humans but seem to primarily affect the nerve cells of insect prey. Both effects probably result from damaged or rearranged cell membranes.
To better understand how spider venom damages cells, my team and I crystallized and took X-rays of a toxin from a Chilean six-eyed sand spider as it binds to target molecules found in cell membranes. We were amazed to behold a structure that reveals how the toxin binds to cell surfaces. Clearly visible in the mouth of the enzyme were the cell surface molecules, positioned in a way that showed how the enzyme cuts the head off and turns it into a ring.
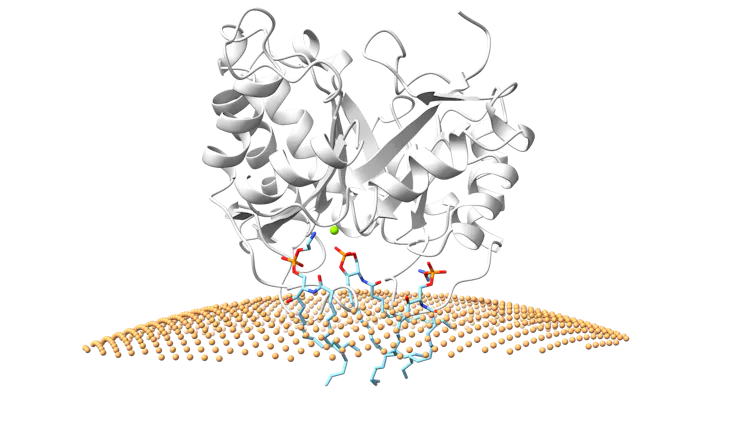
Matthew Cordes/ChimeraX, CC BY-SA
When we compared the structure of the toxin when it’s bound to its target molecules to its structure when it is not, we saw changes that suggest it gets activated when it binds to cell surfaces. That is, it begins to damage cells once it attaches to their surface.
Uncovering the recluse
True to their name, recluses tend to reside in dark, covered places such as woodpiles, closets and pillowcases, and they may accidentally come into contact with people. They are not aggressive, but they do bite when threatened. The most common symptom is a serious skin wound that may require grafts, but the toxin may also damage red blood cells and cause life-threatening kidney failure.
Recluse spider lesions can be misdiagnosed due to their similarity to sores from bacteria such as methicillin-resistant Staphylococcus. There are no approved treatments in the U.S., though antivenoms are available in South America.
Our hope is that this work can guide scientists in developing new ways to treat spider bites and block the effect of their toxins, by either interfering with their ability to bind to the surface of cells or to chemically alter them.
![]()
Matthew Cordes has received funding from the National Science Foundation and the Bio5 Institute.
– ref. How does spider venom damage human cells? Researchers uncover the killer mechanism of recluse spider toxin – https://theconversation.com/how-does-spider-venom-damage-human-cells-researchers-uncover-the-killer-mechanism-of-recluse-spider-toxin-279903
